News
Stay up to date on the latest crypto trends with our expert, in-depth coverage.

1Bitget UEX Daily|US-Iran Talks Renewed Differences, Oil Prices Jump Again; Google in Talks with Marvell to Develop New AI Chips (2026-04-20)2April 20 Financial Morning Brief: U.S. and Iran Accuse Each Other of Attacking Ships, Strait of Hormuz Closed Again, Gold Price Battles at 4750 Level, Oil Price Rebounds Nearly 7%3A "high sensitivity week" arrives for global markets! US-Iran negotiations, Walsh hearings, and a wave of global data set to make an impact

Huatai Futures: US-Iran Negotiations Continue, Precious Metal Prices Rebound
新浪财经·2026/04/15 02:05

B ($B) 24-hour price range 84.8%: abnormal selling surge triggers drastic price swings
Bitget Pulse·2026/04/15 01:59

ZAMA (ZAMA) fluctuates 62.5% in 24 hours: Listing on Korea's Upbit and Bithumb drives price surge
Bitget Pulse·2026/04/15 01:54

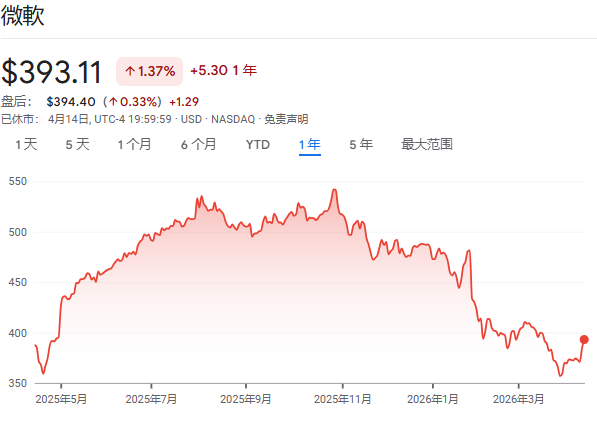
Is it time for Microsoft to "buy the dip"?
华尔街见闻·2026/04/15 01:07

BGSC (BugsCoin) fluctuated by 61.1% in 24 hours: Trading volume surge triggers severe price volatility
Bitget Pulse·2026/04/15 01:07
Flash
20:20
US stock major asset ETFs saw mixed performance; Brent crude oil fund closed up more than 4.3%, while emerging markets ETFs and gold ETFs fell by up to 0.8%.U.S. Treasury 20+ Year ETF, Long Dollar Index, Long Yen, S&P 500 ETF down up to 0.20%, NASDAQ 100 ETF down 0.32%, Emerging Markets ETF down 0.72%, Gold ETF down 0.86%.
20:18
BridgeBio Oncology Therapeutics, Inc recently revealed that the eagerly awaited updated data from its Phase I clinical trial is expected to be released in the second half of 2026.The announcement of this timeline provides clear expectations for investors and industry observers following the company's R&D progress. This data update is crucial for assessing the safety and preliminary efficacy of the relevant candidate drugs. The latest developments in the company's R&D pipeline may influence its future clinical development strategy and market positioning.
20:18
BridgeBio Oncology Therapeutics, Inc. (Bbot) announced that its innovative therapy Bbo-11818, for adult patients with advanced KRAS-mutated pancreatic ductal adenocarcinoma, has been granted Fast Track designation by the U.S. Food and Drug Administration (FDA).This designation will accelerate the development and review process of the candidate drug, bringing new treatment hope to patient groups with urgent unmet medical needs. As the most common type of pancreatic cancer, KRAS-mutant pancreatic ductal adenocarcinoma has long lacked effective targeted therapy options. Obtaining Fast Track designation not only demonstrates the therapeutic potential Bbo-11818 has shown in early research, but also means the drug can benefit from more frequent communication with the FDA during subsequent clinical trials, potentially expediting its progress to the marketing application stage.
News